Neutrons to study the interplay between aquaporins and porous silica in water treatment
Why?
Professor Martin Andersson (Chalmers) -- Access to safe and clean drinking water is one of the basic needs of humanity, an access that is currently threatened by global concerns such as increasing pollution and climate change. The importance of energy efficient water treatment has never been greater, both in small- and large-scale applications. Today, water purification typically includes a size-exclusion based filter that is specialized in removing a certain number of common water pollutants. But these filters do not remove all the pollutants present in water. So why not develop a filter that produces clean water regardless of pollutant? Guess what – nature already did. Aquaporins are proteins present in cell membranes that allow the passage of water molecules only. Unfortunately, it has proven difficult to use these proteins straight away, since it is quite difficult to keep aquaporins stable with time. Using a more robust filter material such as porous silica as a substrate for the proteins could possibly be the solution, but would the protein be satisfied on such a substrate and keep on transporting water?
How?
It is complicated to assess if water channelling proteins are functional in the small-scale setting currently available in the lab. Protein structure and behaviour are therefore used as indicators of function. Neutron reflectivity is an excellent tool to accurately determine the average extension of the intracellular domains of aquaporins situated at a planar surface. By comparing the extension of the protein into the pores with their extension into bulk it is possible to get an estimation on how the proteins are arranged at the interface, which is key to make this kind of filter work. These results were recently published in the scientific journal Nano Letters.

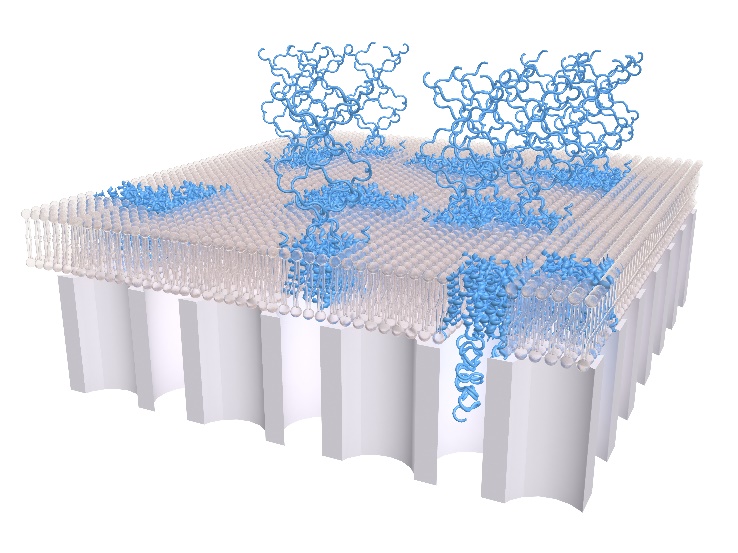
Figure 1. Illustration showing the water filter consisting of silica (grey), aquaporins (blue), and lipids (brown).
Who?
This project is led by Professor Martin Andersson at the department of Chemistry and Chemical Engineering at Chalmers University of Technology in collaboration with Malmö University, University of Gothenburg, Uppsala University, and Los Alamos National Laboratory. Besides the expertise of the collaborating groups, access to leading European neutron facilities such as ISIS Neutron and Muon Soruce in the UK, Institute Laue-Langevin in France, and Heinz Meier-Leibniz Zentrum in Germany is essential to the success of this project. Further use of neutrons within the project has already resulted in more people from various locations getting involved.
What's next?
Besides using neutrons to explore other approaches to stabilise aquaporins, we would like to apply the method to investigate the behaviour of other transmembrane proteins (typically very attractive in pharmacy) supported on porous substrates.
Contact: Professor Martin Andersson
martin.andersson@chalmers.se
Chemistry and Chemical Engineering
Chalmers University of Technology
This project is funded by the Swedish Research Council Formas and the INTERREG Öresund-Kattegat-Skagerrak programme.